|
Photoinhibition, which is largely an impairment of PSII caused by high irradiance, has been demonstrated in coral symbionts during thermal stress with the rate of photoinactivation of the core D1 protein exceeding the rate of repair. Indeed, this initial site of impact remains elusive, with many studies aiming to identify the origin of photosynthetic impairment in Symbiodinium under simulated bleaching conditions. While such results are indicative of impacts to this early stage of the light reactions of photosynthesis, it is not clear whether inhibition of PSII activity is the initial site of damage induced by bleaching conditions, or whether it is a secondary or tertiary impact only detected following inhibition or damage at another site ( Smith et al., 2005). A great deal of evidence has been collected, primarily through non-invasive chlorophyll fluorescence techniques, demonstrating the loss of photosystem II (PSII) photochemical efficiency during a bleaching event ( Jones et al., 1998 Warner et al., 1999 Hill et al., 2004a Hill et al., 2004b). A sustained loss of photosynthetic performance in coral symbionts is a well-defined characteristic of a thermal stress response ( Jones et al., 2000). Coral bleaching events are therefore predicted to become more frequent, widespread and severe with climate change ( Hoegh-Guldberg, 1999).īreakdown in the coral symbiosis has been linked to an initial photosynthetic impairment in the endosymbiotic algae. It is forecast that ocean temperatures will continue to increase with global warming due to anthropogenic emissions of greenhouse gasses ( IPCC, 2007), placing corals more at risk of having sea temperatures exceed their upper thermal maximum. Elevations in sea surface temperature, as small as 1–2☌ above the summer average, in combination with high irradiance, are well known to cause mass coral bleaching events on coral reefs around the world ( Hoegh-Guldberg, 1999). Coral bleaching involves the expulsion of Symbiodinium and/or the loss of photosynthetic pigments from within algal cells, resulting in a paling, or bleaching, of the coral. 
This mutualism is key to the high productivity found on coral reefs, often in oligotrophic, tropical waters, but can be highly sensitive to environmental perturbations, with a breakdown in the symbiosis a common stress response ( Hoegh-Guldberg et al., 2007). Reef-building corals form an endosymbiosis with dinoflagellate algae from the genus Symbiodinium (also known as zooxanthellae), which reside within the endodermal tissue of the cnidarian. These findings do not support the hypothesis that temperature-induced inhibition of the Calvin–Benson cycle alone induces coral bleaching. While KCN is an inhibitor of the Calvin–Benson cycle, it also promotes reactive oxygen species formation, and it is likely that this was the principal agent in the coral bleaching process. In contrast, KCN did activate a bleaching response through symbiont expulsion, which occurred in the presence and absence of thermal stress. 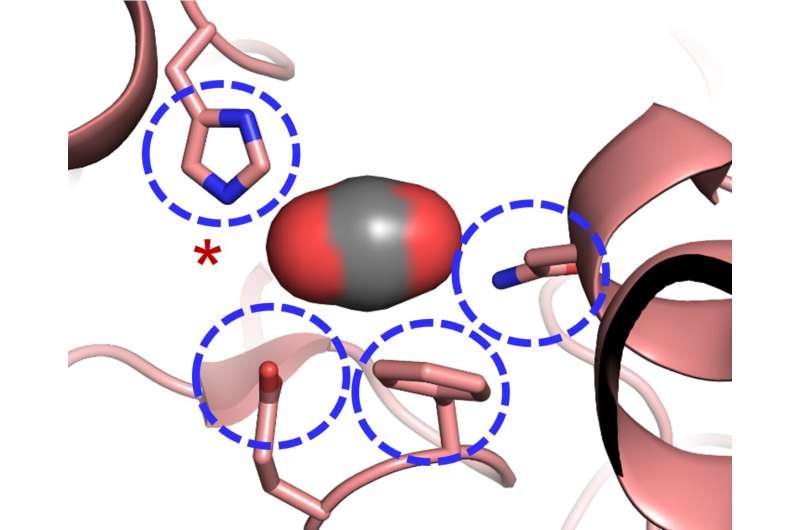
GA did not affect the severity of bleaching, nor induce bleaching in the absence of thermal stress, suggesting inhibition of the Calvin–Benson cycle by GA does not initiate bleaching in P. Both 3 mmol l −1 GA and 20 μmol l −1 KCN caused minimal inhibition of host respiration, but did induce photosynthetic impairment, measured by a loss of photosystem II function and oxygen production. Inhibitor concentration range-finding trials aimed to determine the appropriate concentration to generate inhibition of the Calvin–Benson cycle, but avoid other metabolic impacts to the symbiont and the animal host. Two inhibitors of the Calvin–Benson cycle were used in cultured Symbiodinium cells and in nubbins of the coral Pocillopora damicornis to test the hypothesis that inhibition of the Calvin–Benson cycle triggers coral bleaching.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
